The techniques of imparting color to various crystalline phases preferentially are well known in geology (see Carver, 1971, and Hutchison, 1974). Stain differentiation between plagioclase and potash feldspars and between various carbonate minerals is commonplace, using particles, thin sections, and polished slabs. Stains and etches are those liquids or vapors that, when applied to the polished cross section of a clinker or to a sample of portland cement, preferentially color or dissolve certain phases observed in reflected or transmitted light. The colors mainly result from the refraction, reflection, and interference of light within the thin layer of reaction product formed on the clinker phases. Stains and etches are used to bring out microstructural details of individual crystals. Both stains and etches can be related to the relative reactivities of various clinker phases. Photographs 3-1 through 3-6 illustrate some of the effects of a few stains and etches.Perhaps the most thorough treatment of the subject of stains and etches is the work of Ellson and Weymouth of Australia (1968). Their paper lists approximately 43 reagent solutions and their effects on portland cement and blast furnace slag phases in terms of (a) reaction type (stain or structural etch), (b) time required for the desired effect, (c) recommended temperatures, and (d) concentrations. Futing summarized the application of many varieties of etches in 1986.Much of the information given in this chapter was extracted from the work of John Marlin of the Oklahoma Cement Corporation (now a subsidiary of Lone Star Cement Company, Greenwich, Connecticut). Many of his recipes and results (1978 and 1979) are reproduced in this chapter with only slight modification but only a few have been tried by the present writer. Marlin recommends making fresh solutions every two months for most of these stains and etches. Most of the solutions described in the following pages have simultaneous staining and etching effects, and, unless stated otherwise, the tests are carried out at room temperature. It will be obvious that the effects of various etches and stains are also functions of time and clinker phase composition.Relative reactivities of silicates among several clinker samples, or comparison of the phase percentages of clinkers from different daily productions or different cement companies, can be determined by etching and staining several polished sections simultaneously at the same temperature. To facilitate this technique, one can combine several polished sections with a rubber band, immersing the assemblage in the etchant for the required length of time. Thus all sections are exposed simultaneously for the same length of time, at the same temperature, and relative rates of reaction can be evaluated according to the colors produced. Similar tests can be performed with 0.2% nital and 0.01% aqueous ammonium chloride. CDTA in successive 15-second applications with examinations after each is particularly good to evaluate the relative rates of silicate reactivities in a suite of samples etched simultaneously. Reaction rates can be increased by heating the polished section with the hair dryer for a few seconds prior to application of the etchant.Another helpful procedure in polished section examination is to immerse only one half of the polished surface in water, for example, holding the section with a pair of forceps, spray wash the sample with isopropyl alcohol, dry, and then rotate the sample 90˚ immersing half of the section in nital. Thus the surface is divided by this procedure into quarters: one quarter with only water, one quarter with water plus nital, one quarter with only nital, and a quarter remaining with no etch.
Aluminates and Free Lime
A. Potassium hydroxide—ethyl alcohol solution(5%) is placed in contact with the polished section for no more than 20 seconds. Wash the section surface in a 1:1 ethyl alcohol-water solution followed by a wash in isopropyl alcohol, and buff for approximately 15 seconds on MicroclothTM *,** wetted with isopropyl alcohol. Wash with isopropyl alcohol. C3A turns blue.B. Sodium hydroxide—ethyl alcohol solution is prepared with 2.5 g of sodium hydroxide plus 40 mL of water plus 10 mL of ethyl alcohol. If the contact of the polished surface with the solution is more than roughly 20 seconds, a deposit from a reaction between hydroxide and aluminate forms that buffing will not remove. C3A turns blue. If determination of alkali sulfates is desired, stain only one time for approximately 10 seconds, washing with 1:1 ethyl alcohol-water solution, followed by isopropyl alcohol. Do not buff. This treatment will darken alkali sulfates slightly and with prolonged treatment (as for C3A) will dissolve the alkali sulfate, producing a dark void.C. Potassium hydroxide solution (0.1 molar aqueous) can be applied in single drop fashion or in a small puddle on a polished surface for 30 seconds. Rinse with an isopropyl alcohol spray and dry with forced warm air. C3A and alkalialuminate stain blue-brown, alkali sulfate darkens, and free lime turns brown.D. Boiling sodium hydroxide solution (10% by mass) will turn calcium aluminate blue or brown in 20 seconds in a high-alumina cement. Etching 30 seconds with a 1% borax solution turns C12A7 gray (Long, 1983).E. Warm distilled water (40°C) in 5 to 10 seconds turns aluminates blue to brown, alite light tan, free lime multicolored, and does not affect belite.SILICATESA. Dilute salicylic acid stain is mixed as follows: 0.2 g salicylic acid plus 25 mL of ethyl alcohol plus 25 mL of water. After a 20- to 30-second immersion, followed by an alcohol spray wash, alite and belite are blue-green. A modification of this stain is 0.2 g of salicylic acid plus 25 mL of isopropyl alcohol plus 25 mL of water, which, after 20 to 30 seconds, reveals that alite stains 50 percent faster than belite and which, therefore, can be used to distinguish the two phases. A precise immersion time for a series of samples aids in their comparison.
B. Salicylic acid etchant is made by dissolving 0.5 g of salicylic acid in 50 mL of methyl alcohol. After a 45-second etch the alite and belite areclearly seen, the latter showing its lamellar structure. Longer contact with the solution degrades belite lamellae. Alite is more strongly etched than belite. This etchant can be used prior to ammonium nitrate for alite-belite differentiation with very little effect on the matrix phases. Reaction of salicylic acid in ethyl alcohol is 50 percent that of methyl alcohol and attacks alite about twice as fast as belite. With isopropyl alcohol, however, the reaction is less than 25 percent that of methyl alcohol, and alite is intensely and rapidly attacked, with belite almost nonreactive.C. Nital is perhaps the most common etchant and stain for silicates and improves with age. Nital is 1.5 mL of nitric acid (HNO3) in 100 mL of ethyl, methyl, isopropyl, or amyl alcohol. The author routinely uses a solution of 1 mL of HNO3 and 99 mL of anhydrous isopropyl alcohol. The solution quickly reacts in 6 to 10 seconds with alite and belite. At a 0.05% dilution the reaction time is 20 to 40 seconds. Ono (1995) relates alite reactivity to color produced with 0.2% nital. Depending on the relative reactivity of silicates, alite normally turns blue to green, belite is brown to blue—both silicates showing details of internal structure. Nital superimposed on a 20-second potassium hydroxide etch turns C3A light brown and colors the silicates.D. Acetone-water solution (in a 1:1 proportion) can be used as a rinse because it reacts slowly on silicates. A 120-second stain time reveals wellstained alite and belite. C3A is also visible.
E. Isopropyl alcohol solution (10%) is an easily made stain (10 mL of isopropyl alcohol plus 90 mL of water) that reacts strongly with alite and weakly with belite in 30 seconds to 2 minutes.C3A exhibits a weak reaction. Compare with HF vapor.F. Maleic acid attacks alite and belite at about equal rates and a little faster than salicylic acid. When followed by NH4NO3, it does not give color distinction to alite and belite.G. Ammonium chloride (saturated, aqueous) colors a hexagonal section of alite (perpendicular to the threefold crystallographic axis) light yellow. The slender hexagonal section of alite (parallel to thec axis) is colored blue. Zoned crystals in the slender hexagonal section show light-blue cores and dark-blue rims. Ono (1995) recommends anaqueous ammonium chloride solution (0.2 to 2.0%) for etching of polished sections. He related the thickness of the film produced by etching to the color of the resulting reflected light with the equation R = 2d(n), where R is retardation, d is the thickness of the thin film of etching product, and n is approximately 1.5. Thus R = approximately 3d. A table of etch colors is presented in relation to different values of R and d, using a well-burnt clinker and 0.5% ammonium chloride. Likewise, alite etch colors produced with 0.2% HNO 3-alcohol are presented in relation to location in the clinker, R, and d. Many of Ono’s photomicrographs, however, indicate etching for 20 seconds with water followed by 5 seconds with 2% aqueous ammonium chloride.Uchikawa (1992) summarized the quality-control techniques for cement and concrete and presented a numerical etch-color scale from 0 to 16, relating each clinker phase reactivity to etch color, using 0.01% aqueous ammonium chloride. The interpreted reactivities were said to be relevant to the initial and early stages of hydration, as well as the sintering conditions. Alite was reported to be more easily etched with “the increase in heating rate, the decrease in burning temperature, the coarsening of the particles of raw materials, and the burning atmosphere approaching reducing.” Interstitial microstructure (ferrite and aluminate crystal sizes, and ferrite crystal shape factor) and the etch colors of alite were correlated with heat of hydration, mortar flow, and setting time. Relatively slowly cooled matrix was hydraulically more reactive but led to lower, more variable, mortar flow and lower fluidity. The more easily a clinker was etched, the shorter the initial setting time, which was also shortened by 40 minutes when free lime was increased by only 0.5%. Slowly cooled belite (Type IIIb variety showing extended lamellae and remelting) was shown to be colorless and, on a color basis, indistinguishable from quickly quenched belite. Alite with high amounts of impurities and high Al2O3/Fe2O3 ratio correlated with low 28-day strength. Under reducing conditions, triclinic alite and partial transformation of belite to the gamma polymorph were produced, along with smaller alite, larger belite, and lower strength development.Dorn and Adams (1983) have described the various etch rates of alite and belite in relation to hydraulic activity. A blue color on alite after a 15- second nital etch was said to represent an active alite.H. Another variety of theammonium chloride stain is made as follows:1 g NH4Cl + 20 mL H2O+ 20 mL ethyl alcohol + 10 mL acetone + 150 mL isopropyl alcoholEffects of this stain are very similar to those of NH 4NO3 except the NH4Cl stain is approximately 25% faster. Alite turns brown in 10 to 20 seconds; belite is unaffected. This stain can be used directly as a belite indicator by extending the submersion time to 30 to 45 seconds. Alite turns yellow to yellowish green and belite to brown. The effects of this NH 4Cl solution are not as clear for belite lamellae as NH 4NO3 following salicylic acid.I. Ammonium nitrate solution is composed of the following ingredients:1 g NH4NO3 + 20 mL H2O + 20 mL ethyl alcohol+ 10 mL acetone + 150 mL isopropyl alcohol Alite is colored in 25 to 30 seconds. With increasing treatment time, the colors on the silicates progressively range from light brown to brown to purplish brown to blue to blue-green to green to yellow-green. Normally, when alite is stained yellow-green, belite will be brown. This solution can be applied following the salicylic acid stain to show alite and belite with an approximately 30- second submersion time.J. Hydrofluoric acid (HF) vapor, used to etch and stain a polished clinker, has been a very informative technique (Long, 1982a). Almost all the clinker phases can be differentiated with an HF vapor etch. The HF is kept at a temperature of 20°C to 22°C. A finely polished surface is held for 5 to 10 seconds in HF vapor and, after waiting a minute or two for the excess HF fumes to leave the polished surface, the section is examined in reflected light. Belite turns blue and alite is brown. With practice at varying the etch times one can develop reliable HF-vapor etch criteria for other phases such as the alkali sulfates. Prout reported (personal communication, 1984) that a temperature differential between fume and specimen enhances etching. The specimen can be cooled or the HF warmed. Incidentally, C2AS (melilite) is colored with HF vapor and occurs in high-alumina cement (Long, 1983).NOTE: Care must be taken to avoid damaging the microscope objective lens with HF vapors emanating from a freshly etched polished section. Waiting a few minutes before examination is recommended. Because of the extreme danger in skin contact with HF, suitable precautions with gloves and ventilated hood are strongly advised.K. Distilled water was described by Brown (1948) as an etch that enabled one to discriminate nine clinker phases after a relief polish. With the use of present-day materials and equipment, Brown’s procedure is as follows: (1) Final polish on Microcloth™ or nylon with 0.05 µm alumina. (2) A removal etch, using distilled water at pH 6.8 to 7.0, is developed by holding the polished surface with moderate pressure on a rotating saturated Microcloth™ for two to three seconds while the distilled water is poured onto the Microcloth™. (3) Wash quickly with isopropyl alcohol and dry with forced warm air. Periclase remains topographically high due to its relative hardness. Free lime etches dark to iridescent green and blue. C 3A turns dark blue. What Brown called “dark prismatic” (actually, alkali aluminate) and ragged dark interstitial material turn faint blue. Alite becomes brown, and belite is recognized morphologically. Alkali sulfates are dark.
EXAMINATION OF STAINED CEMENT
A few milligrams of cement or crushed clinker are immersed in a puddle of the stain solution in a small beaker for 1 minute, after which the mixture is washed with isopropyl alcohol into a 75-µm (No. 200) sieve, and finally into a watch glass. Excess alcohol in the watch glass can be drawn off with a paper towel. The residue is slowly dried in the watch glass at a temperature of approximately 40°C, under a heat lamp or on the slide warmer, and examined with oblique or transmitted light on the stage of a polarized-light microscope. Oblique light illuminates the particles from a point beside the microscope objective. Sulfate minerals retain a prominent pink to red color. A white paper background accentuates the color contrast while viewing the stained particles with oblique lighting. Refractive-index oils as mounting media can be used with transmitted light; however, the alteration of some sulfates during the staining may interfere with the refractive-index determinations.The following easy method for concentrating some of the sulfates in cement or crushed clinker for microscopical examination has been developed. (1) Place a small portion of sample into a watch glass and flood with isopropyl alcohol. Swirl the mixture in the watch glass for a few seconds to concentrate the white-to-clear sulfate particles in the center. (2) Draw the liquid off with a paper towel and dry the remaining powder in the watch glass under a heat lamp or on the slide warmer (no hotter than 40°C). (3) Using a thin metal spatula, scrape off the top-central area of the residue in the watch glass and place it on a microscope slide for examination in oil with a selected index of refraction (n).Although the mount is impure (contains several phases), in an oil with a refractive index of 1.54, gypsum (n = 1.52 to 1.53) and plaster (n = 1.55 to 1.57) can be distinguished. Gypsum has inclined extinction, but the extinction of plaster is straight (Lea, 1970). Anhydrite (n = 1.57 to 1.61) also shows straight extinction, cleavages at right angles, and relatively high birefringence. Extinction angles to differentiate gypsum from plaster, however, are difficult to apply due to the very finely microcrystalline structure common in gypsum and plaster particles. This microstructure appears to be a product of recrystallization due to relatively high temperature, grinding stress, or both, which convert the previously continuous atomic structure into a myriad of minute polygonal crystalline units. Dorn (personal communication, 1985) reported that sulfates such as gypsum and plaster float in refractive-index oil (n = 1.71) and can be skimmed aside for study in a powder mount; anhydrite may sink in the liquid. A simple heavy liquid-centrifuge method appears to be an efficient technique for separation of cement sulfates for microscopical examinations and x-ray diffraction, leading, perhaps, to a quantitative determination of phase abundance.Gypsum, plaster, alkali sulfates, and epezite float in refractive index liquid (n=1.715) and accumulate just beneath the cover glass
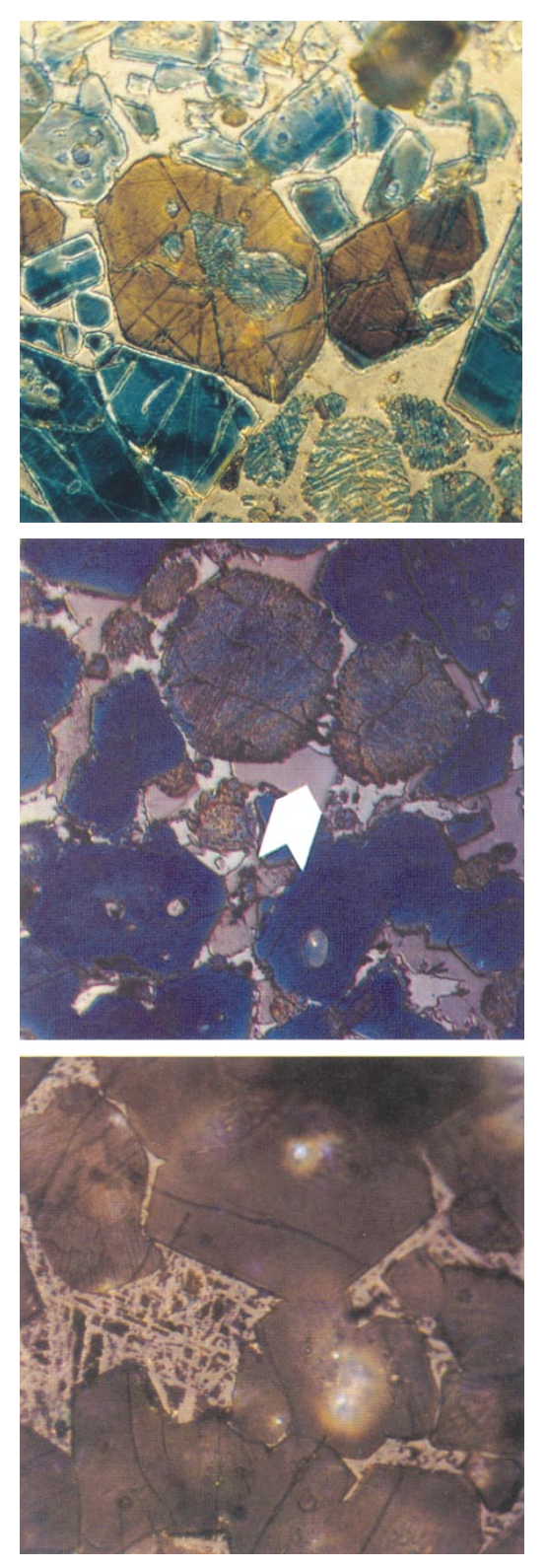
PHOTOMICROGRAPHS OF EFFECTS OF STAINS AND ETCHESPhotograph 3-1 Coarsely crystalline clinker from coarse seashell feed. Angular subhedral to euhedral blue alite, round tan-to-brown belite with wide lamellae, and matrix of dark aluminate (C3A) and brightly reflecting ferrite (C4AF). Coal- and coke-fired, semidry process kiln, 1850 tons/day. High maximum temperature, long burning time, slow heating rate, quick to moderately quick cooling, 44.1 MPa. (S#A6610)Polished section Nital on KOH etchField dimensions = 0.21x0.21 mmPhotograph 3-2 Blue coloration on alite with an otherwise uniform tan color on alite crystals. Possible explanations include differences in crystallographic orientation, chemical composition, structural state (for example, monoclinic versus triclinic), or perhaps combinations of these. (S#A6612)Polished section Nital etchField dimensions = 0.21x0.21 mmPhotograph 3-3 Differential coloration by nital etch on angular alite and round belite. Coarsely crystalline clinker from coarse raw feed. Coal- and coke-fired, semidry process kiln, 1850 tons/day. High maximum temperature, long burning time, slow heating rate, quick to moderately quick cooling. (S#A6613)Polished sectionField dimensions = 0.21x0.21 mm

PHOTOMICROGRAPHS OF EFFECTS OF STAINS AND ETCHES (CONTINUED)Photograph 3-4 Aqueous NH4Cl etch (30 to 40 seconds). Sections perpendicular to the c crystallographic (three-fold axis) of alite are brown. Sections parallel to c are blue. (S#A6614)Polished sectionField dimensions = 0.12x0.12 mmPhotograph 3-5 Large, round, slightly ragged belite; blue subhedral to anhedral alite; and matrix of aluminate (C3A, arrow) and ferrite. Superimposed nital over KOH produces increased clarity in matrix phase definition. (S#A6615)Polished section Reflected light with stopField dimensions = 0.21x0.21 mmPhotograph 3-6 Well-differentiated, finely microcrystalline matrix of aluminate (C3A) and ferrite (C4AF) in nital-etched polished section. (S#A6611)Oil immersion Reflected lightField dimensions = 0.10x0.10 mm