Introduction
Sulphur is probably the “easiest” pollutant from a gas clean-up point of view. A first reason for that is certainly that the chemistry involved is relatively simple and (hence) well understood. In combustion plants sulphur in fuel will eventually be oxidised to sulphur dioxide SO2 and some sulphur trioxide SO3, be it that small amounts may be bound by ash-forming minerals as sulphates. At the same time, a long history of SO2 capture with limestone and other calcareous rocks exists, which is accomplished at relatively low costs due to the abundance of these materials in nature. Hence, it is no surprise that in response to stricter legislations introduced during the 1970-1980s, significant sulphur oxides (“SOx”) emission reductions of were rapidly implemented (Ponting, 1991). In Europe, sulphur emissions dropped by 53% since 1980 (Acid News, 2000a). This in contrast with the ongoing struggle to reduce nitrogen oxides (NOx) emissions from the same facilities.In gasification or pyrolysis plants hydrogen sulphide H2S will be the most important sulphur compound. Methods for its control have been developed first by the petrochemical industry, while later methods based on (regenerable) sorbents were specially developed for coal gasification units. Upon release to the environment, H2S will be oxidised to SO2.One fact remaining, though, is that sulphur must eventually be disposed of in some chemical form. Unlike fuel-related nitrogen, that without any environmental concern may be released partly as molecular nitrogen N2, there is no such “escape route” for sulphur. Fuel-related sulphur that is captured may end up as molecular sulphur, sulphuric acid, or bound in flue gas desulphurisation (FGD) residues like gypsum, and partly in bottom ashes and fly ashes. In other cases, such as cement kilns, the cement product may contain a significant amount of the sulphur from the fuels used (coals, petcoke, alternative fuels) without causing problems.For utilities and power plants firing sulphur-containing solid fuels SO2 capture can be implemented with guaranteed success, but a different situation exists for the fuels derived from oil. As a result of tight limits on sulphur contents of transport fuels like gasoline and light fuel oils, it is difficult for the petro-chemical industry to find an outlet for the sulphur that is obtained from oil refining. Currently much of that
sulphur is found in heavy fuel oils used on ships without emissions control, that is, at those seawaters where high-sulphur fuels for shipping are accepted ("Cleaner Shipping", 1997) (L section 3.15).Sulphur emissions to the environment may be divided in being the result of human activities, basically the combustion of fuels for heat and power or transport, plus a contribution from natural sources. Volcanoes emit significant amounts of sulphur as well. Emissions of sulphur dioxide are correlated with deposition of sulphur compounds, often in the form of dilute sulphuric acid, part of which is known as “acid rain”. Figures 3.1 and 3.2 give an impression of the situation in Europe during the last decade. It is seen that the sulphur emissions result in deposition mainly in the densely populated and industrialised “band” from England via Germany to Central and South-East Europe. The countries in Scandinavia can be considered as importers of sulphur-related pollution, whilst countries like Spain, Portugal, Ireland and Russia appear to export most of their sulphurous pollutants
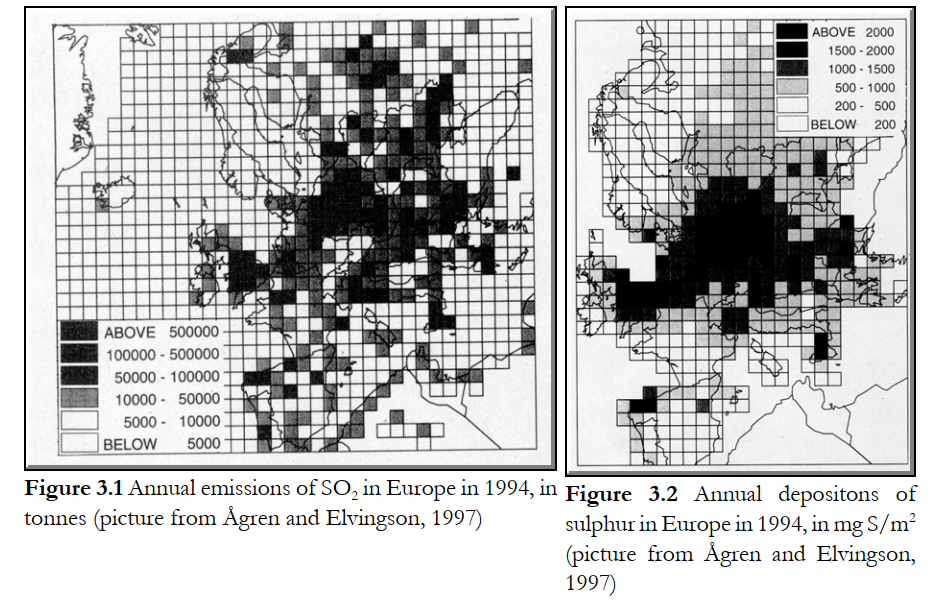
Reasons for controlling SO2 emissions are related to its contribution to acid rain, and more recently it was recognised that sulphate-containing aerosols form a health hazard as well, causing problems to the respiratory and cardio-vascular systems. With oil and gas processing, and gasification processes H2S is the most important sulphur species
to be controlled, being not only highly poisonous and bad smelling, but also resulting in SO 2 after oxidation in the environment.In this chapter, the routes from sulphur present in fuels to pollutants and methods to avoid these from being emitted to the atmosphere are discussed. After a short discussion on in what forms sulphur occurs in fuels, the essentials of the chemistry of sulphur during combustion and gasification are given. This is followed by some emission standards for SO2, for Finland i.e. the EU and some others. A general summary of approaches to sulphur control is given, which is followed by a rather extensive treatment of the most important technique: flue gas desulphurisation (FGD) for SO 2. Wet, semi-dry and dry FGD with non-regenerable or regenerable sorbents are discussed. High temperature SO2 capture is discussed addressing furnace sorbent injection and in situ SO2 control during fluidised bed combustion (FBC). After this the point of focus switches to H2S. First, low temperature processes for H2S removal from gases are considered, followed by the famous Claus process. Methods based on H 2S capture with calcium-based and regenerable sorbents as used in relation to (coal) gasification, sometimes refered to as high temperature gas clean-up for sulphur, are discussed next. Finally, the control of sulphur compounds emissions from transport vehicles is addressed
.
Sulphur in fuel
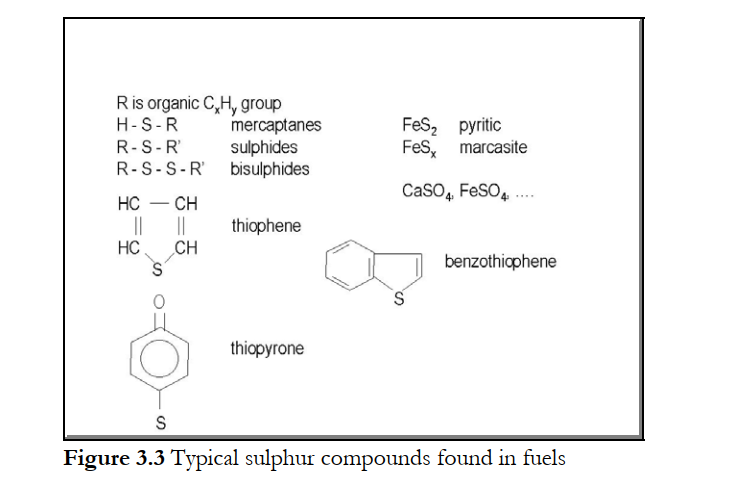
In solid fossil fuels, such as coals ranking from brown coals and lignites up to anthracite, peat, oil shales etc., sulphur is present in two inorganic forms, being pyritic sulphur (FeS2), and sulphates (Na2SO4, CaSO4, FeSO4) and as organic sulphur. Organic sulphur is bound in these fuels as sulphides, mercaptanes, bisulphides, thiophenes, thiopyrones etc., see Figure 3.3.These organic compounds are also found in unrefined oil and heavy fuel oils. In natural gases as well as in raw fuel gases from gasification processes sulphur is generally present as hydrogen sulphide (H2S) and carbonyl sulphide (COS).Biomass fuels such as waste wood, straw, bagasse (residual from sugar cane processing) and energy crops (Salix, Miscanthus, etc.) contain very small amounts of sulphur. The relatively large amount of sulphur in bark is apparently correlated with the exposure of this part of the tree to the surrounding atmosphere and occasional acid rain and other downfall. In waste-derived fuels, sulphur can be found in car-tyre scrap and electronic scrap as a vulcaniser, in food-derived wastes (onions and garlic) etc. Specific for pulp and paper processing is black liquor, for which the large sulphur
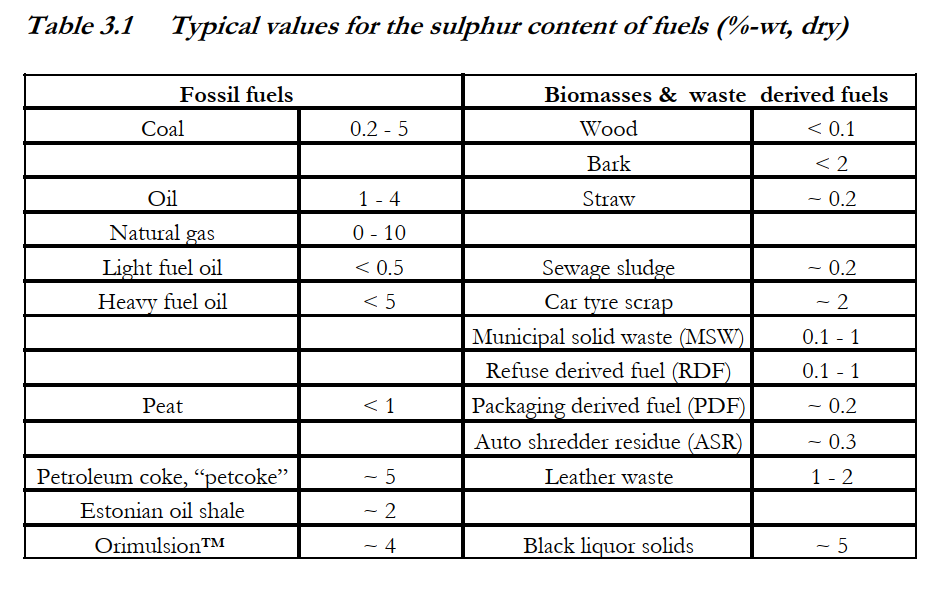
content, mainly as Na2SO4, is recovered in reduced form as Na2S in so-called recovery boilers (Hupa, 1993). Typical values for the sulphur content of various fuels are given in Table 3.1.
| Formation of sulphur compounds during combustion and gasification |
| In short, the formation of sulphuric pollutants during combustion and gasification can |
| be described by the following chemistry : |

The reactions involving O2 lead to the major sulphuric product during combustion, SO 2 via (R3-2) and (R3-5). Although the thermodynamics of (R3-10) are in favour of SO 3, the oxidation of SO2 to SO3 is a slow chemical reaction. It requires temperatures above 1100EC if it is to proceed as a gas phase reaction, or a catalyst such as oxides of vanadium, nickel and iron for the reaction to become important already at temperatures as low as 500EC.This catalytic effect explains why during the early 20th century when oil-derived transport fuels were introduced corrosion problems arose that were not seen that strongly with coal. The sulphur in the transport fuels was oxidised to SO2 and further to SO 3, catalysed by traces of metallic elements in the fuel, which did not occur that strongly with coal. Upon cooling, this SO3 which had bound with water to form H 2SO4, formed highly corrosive liquid sulphuric acid. The “dew point” of H2SO4, i.e. the temperature at which condensate is formed is determined by the concentrations of water and SO3 in the gas. Figure 3.4 (Backman et al., 1983) gives the dewpoint of sulphuric acid.
For gasification processes the situation is more complicated. The most important sulphur species will be H2S, which is in equilibrium with certain amounts of COS

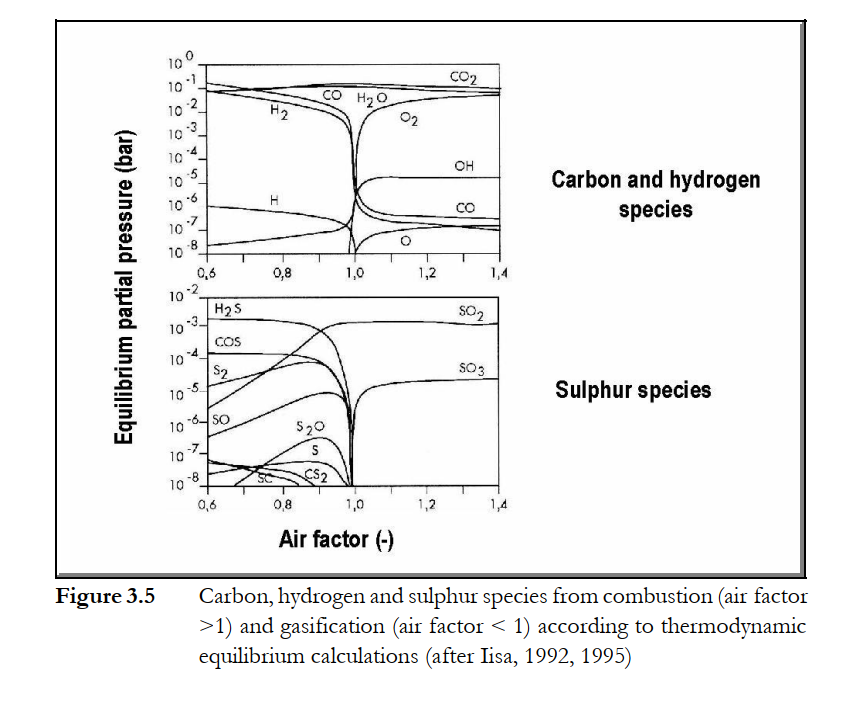
(carbonyl sulphide), CS2 and elemental sulphur Sx (with S8 as a common form) via reactions (R3-6,7,8,9). H2S control is one option here, besides removing the sulphur as SO 2 after an oxidation if that occurs downstream in the process, via reaction (R3-5). Figure 3.5 gives some results of thermodynamic equilibrium calculations for sulphur compounds in combustion (air factor$1) and gasification (0<air factor<1) reactors (Iisa, 1992, 1995). As mentioned above, the SO3 levels shown generally do not occur due to slow SO2 oxidation kinetics, leading to slightly higher SO2 levels than what thermodynamics indicate.
Options for sulphur emissions control
Several routes are open for reducing the sulphur emissions from thermal processes that fire or treat sulphur-containing fuels or materials.
Substitute the fuel
It is obvious that replacing a high-sulphur fuel with a low-sulphur or sulphur-free fuel is an option that should never be forgotten. This depends on the fuel-flexibility of the unit, the economics of operation with another fuel and the adjustments that may be needed, and the contracts with the fuel (or whatever is thermally treated) supplier. It must be noted that a fuel switch may have more benefits, for instance less ash formation or lower taxation when e.g. a renewable fuel is chosen. At the same time, it should not be forgotten that sulphur may have positive effects on ash behaviour in boilers and furnaces, and on the performance of an electrostatic precipitator (ESP) for dust emissions control (L Chapter 5). In some cases, fuel substitutions may also be enforced by local authorities.
Fuel cleaning
A second method that prevents the sulphur from entering the facility is to apply fuel pre-treatment. Inorganic fuel sulphur, especially pyritic sulphur, FeS2, can be relatively easily removed to up to 90% by physical methods based on gravity, flotation, agglomeration or flocculation. Chemical methods may be able to remove organic fuel sulphur as well, involving treatment with alkaline or caustic solutions, oxidative leaching or chlorinolysis with chlorine-based chemicals. The price for these chemical methods is that part of the material is oxidised, which reduces its potential for use as a fuel.
A third method is biological treatment, capable of removing inorganic a well as organic sulphur. It was found that Thiobacillus ferrooxidans converts FeS2 into watersoluble FeSO4,whilst a mutant of Pseudomonas, called CB1 (coal bug 1) consumes organic sulphur in thiophene groups. These techniques may require very small particles, are time-consuming, and may need close process control. All methods mentioned are quite expensive and still may have a limited effect. On the other hand many low-grade coals are “washed” in order to reduce their mineral content which results in some level of sulphur removal as well. A discussion on coal desulphurisation was given by Celik and Somasundran (1994). In a recent paper by Rubiera et al. (1999) it was argued that biological treatment does not have a substantial effect on the combustion behaviour of coal.
Desulphurisation of petroleum fractions has been successfully applied for a long time in the field of petro-chemicals. Using H 2 and a Co/Mo catalyst, sulphur is removed from petroleum fractions by conversion to H2S. Examples of “hydrocracking” reactions are given in Figure 3.6 (van den Berg and de Jong, 1980)

Abatement techniques
Since eventually the sulphur that enters the facility will come out in some form, these methods are based on lowering the sulphur input, with the fuel, to the process. This implies increasing the thermal efficiency of the fuel conversion process by increasing the steam parameters (supercritical or ultra-supercritical steam). Alternatively, the facility can be upgraded by “repowering” - examples are: transforming a pulverised coal fired unit to a (pressurised) fluidised bed unit, implementing combined-cycle technology or even the use of fuel cells (7 Chapter 2). In practically all cases this implies rather expensive changes with many risks.
End-of-pipe control technologies
The last but most widespread option is to remove the sulphur as SO2 or H2S after it has been formed in the process. In fluidised bed reactors, sulphur can be captured in situ in the bed by a calcium-based sorbent (L section 3.11) - for older plants existing
flue gas desulphurisation equipment may become obsolete after repowering to a fluidised bed furnace. The rest of this chapter will concentrate on techniques that accomplish this end-of-pipe control. In all cases this brings with it the production of significant amounts of by-products, such as spent lime and dolomite, gypsum, elemental sulphur or sulphuric acid.