Microscopical descriptions of clinker phases had their origins in 1887 with the work of the French chemist LeChatelier. Following the methods of microscopical analysis of rocks developed by the English geologist H.C. Sorby, founder of petrography and metallography, LeChatelier reported the presence of the following constituents in a portland cement clinker thin section:1. Clear, colorless, angular crystals with a low birefringence, identified as tricalcium silicate
2. Rounded, turbid, yellowish crystals with moderate birefringence, identified as dicalcium silicate3. A dark brown intermediate substance of irregular and ragged form with a lime-iron-aluminate composition (later shown to be calcium aluminoferrite)4. Another material, which, he inferred chemically, should be tricalcium aluminate.
Although it is not clear whether LeChatelier examined cement made by Joseph Aspdin, who patented portland cement in England in 1824, a few comments on the nature of the Aspdin cement appear relevant to the history of clinker microscopy.In 1978 a sample of hardened paste was given to the writer by Norman Gregg of R. H. Harry Stanger, Ltd., Hertfordshire, United Kingdom. Gregg reported that the paste represented several barrels of cement (made by William Aspdin, son of Joseph Aspdin) that had been aboard a ship that sank in the River Thames in 1848 near Sheerness, Kent, England. The story of these barrels of cement and other early cements is told by Blezard (1984).A polished thin section of the hardened Aspdin paste (Photographs 1-1 through 1-4) was examined by the writer and found to contain approximately 10 percent unhydrated portland cement clinker (UPC)
PHOTOMICROGRAPHS OF ASPDIN PASTEPhotograph 1-1 Portland cement clinker particle in Aspdin paste. Subhedral to euhedral pale-green alite; raggedy, round multicolored belite; coarsely crystalline brightly reflecting ferrite; and gray aluminate (left center). Edge of particle shows pseudomorphic hydration effects. (S#A6606)Polished section*KOH followed by nital etchFD (Field Dimensions) = 0.21x0.21 mm

PHOTOMICROGRAPHS OF ASPDIN PASTEPhotograph 1-2 Unhydrated portland cement clinker (UPC) in Aspdin paste. Large, blue-green, angular alite; small, tan-orange round belite (Type II, Insley); brightly reflecting ferrite; and pinkish-gray aluminate, presumably C3A. (S#A6607)Polished section Nital etchFD = 0.21x0.21 mmPhotograph 1-3 Unusually large belite in UPC in Aspdin paste. Note prominent lamellar extensions into ferrite matrix. Probably an effect of CaO resorption during slow cooling. (S#A6608)Polished section Nital etchFD = 0.21x0.21 mmPhotograph 1-4 UPC in Aspdin paste. Water etch reveals dark-blue, coarsely crystalline aluminate, presumably C3A. (S#A6609)Polished section FD = 0.21x0.21 mm
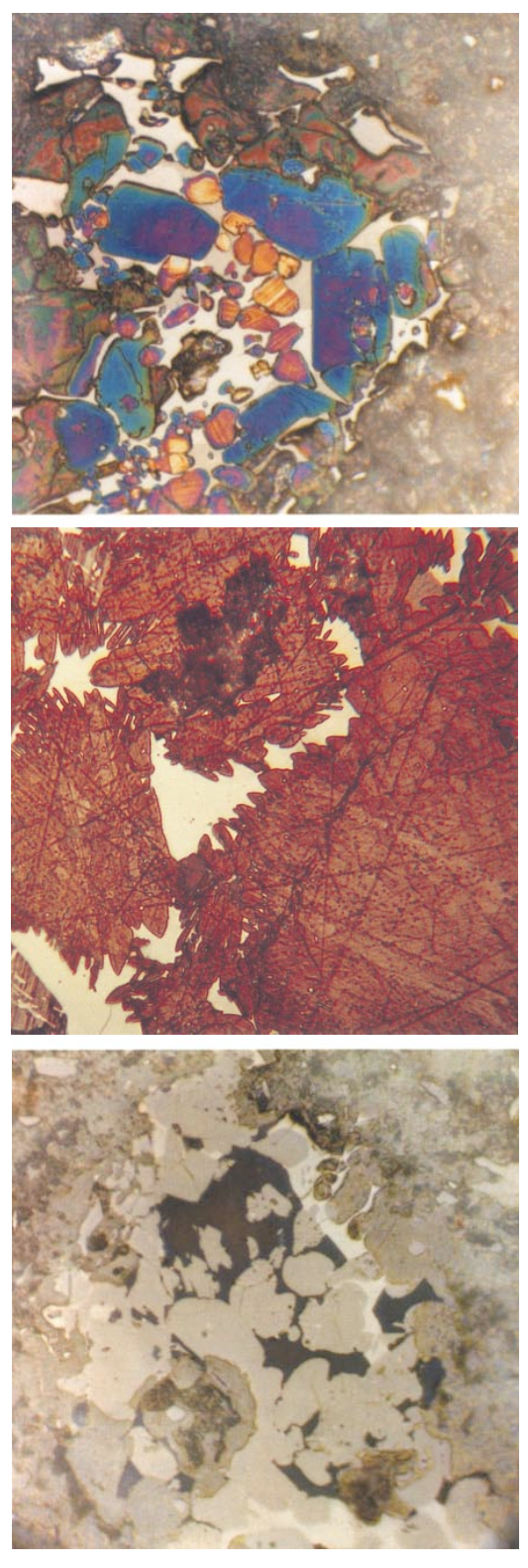
particles. Although the UPCs are far from identical to those of modern production, they clearly contain, among other phases, the four principal phases typical of portland cement (alite, belite, aluminate, and ferrite). Glassy particles were also observed in the Aspdin paste and appear similar to those described by Idorn and Thaulow (1983), who described some of the microscopic characteristics of a precast concrete wall placed in front of Portland Hall, Gravesend, Kent, England, in 1847. The wall is said to have been built for William Aspdin. Further discussion of this wall and the nature of the UPCs is given by Blezard (1981 and 1984), who shows photomicrographs suggesting a coarsely crystalline clinker that was slowly heated and slowly cooled. Cements similar to this Aspdin cement may have comprised some of the samples studied by LeChatelier and other early workers in clinker microscopy.Scrivener (1988) studied the Aspdin paste with backscattered electron imaging (BSE), showing clearly the development of hydration products pseudomorphic after the original clinker crystals and drawing attention to the occurrence of layers of hydration product (“inner product”).In 1897 Törnebohm, a Swedish investigator, possibly realizing that because of compositional variation mineral names might be better suited for clinker phases than chemical formulas, clearly described the optical features of the principal clinker phases in thin sections and powder mounts and coined the terms alite, belite, celite, felite, and also glassy residue. Törnebohm stated that “belite”has two or three sets of cross striations and “felite” has one set of parallel striations formed at low temperature. Törnebohm related microscopical data to burning conditions, stating:1. Well-burned clinkers are less porous and contain better-crystallized colorless alite and dirtygreen to muddy belite.2. Brownish-orange celite functions as a flux, promoting the development of the silicates.3. Underburned clinker disintegrates because of atmospheric moisture combining with residual lime.Törnebohm also made notable contributions to the microscopical understanding of cement hydration, a topic that must be left for future discussion.Richardson (1903-1905) summarized theories on the chemistry of portland cement and demonstrated the use of a polarized-light microscope in the prediction of cement quality from clinker examinations. Richardson stated: “If the structure is coarser and the elements are more segregated, the cement from sucha clinker will be less reliable.” Most of Richardson’s work, however, was in the laboratory where, with numerous sintering experiments, he made frequent use of powder mounts and thin sections to study the products. Richardson, undoubtedly, developed an extensive and systematic body of knowledge that formed foundation for later work by others.Bates in 1912, describing some of the cement chemistry work at the National Bureau of Standards, stated (p. 369) “It was recognized from the first, that in order that the studies, which were to be made, might be complete, a petrographer with a complete outfit for petrographic studies must be installed. All burns would then be examined for their constitution according to the most approved and exacting methods.”Rankin and Wright (1915), although they were not particularly concerned with the interpretation of burning conditions, firmly established the optical properties of pure compounds and the principal phases in commercial cements. These authors systematically investigated approximately 1000 combinations of lime, alumina, and silica with fully 7000 heat treatments and microscopical examinations.Using Törnebohm’s classification, Guttmann and Gille in 1928 tabulated the basic optical properties of clinker phases and the common hydration products. In 1931, Guttmann and Gille summarized the 50-year controversy over the nature of alite and demonstrated conclusively that alite is C3S. *According to Insley (1936), polished section examination of portland cement clinker was reported by Stern (1908) and by Wetzel (1913); but, largely due to poor technique, the metallographic method was abandoned until Tavasci’s very detailed paper in 1934 in which reflected-light microscopy was combined with that from transmitted light.Tavasci (1934) believed that clinker was composed primarily of alite, belite (alpha and beta), and celite (a fine mixture of 3CaO•Al2O3 and 4CaO•Al 2O3•Fe2O3), with free lime as a frequent additional phase. With a series of etches, including nital, oxalic acid, hydrofluoric acid, water, and others, Tavasci carefully described the various effects of these solutions on clinker phases and other synthetic compounds. Tavasci presented rather meticulous descriptions of the forms of belite, suggesting a martensitetype separation in the transformation of alpha to beta.Tavasci classified belite into three morphological types: I, II, and III. Belite I was said to show striations, sometimes like twinning, prevalently in two directions. The striae were described as being relatively thick but not very “fitte” (a term believed by the present writer to mean “etching resistance”). Upon etching with alcoholic nitric acid (nital) the striae were hollowed out with respect to the rest of the grain. Belite II crystals were generally large, containing very “fitte” striae, the dark striae showing relatively less attack by alcoholic nitric acid than the bright striae. Belite III was comparatively small and appeared to be an external zone over a core formed by belite I or II. Coarse striation did not occur in belite III, but fine parallel striations were observed like those in belite II. Belite III was said to contain “a kind of veining formed by inclusions which at high magnification appear white and in strong relief.”In 1936 Insley (about whom much more is said later) clearly showed that (1) alite is tricalcium silicate (C3S), (2) two different habits of dicalcium silicate (C2S) comprise belite and felite, and (3) celite is tetracalcium aluminoferrite (C4AF). Insley’s descriptions and illustrations of clinker phases remain the basis for much of succeeding publications by others.Among the many historically important contributors to the microscopy of portland cement clinker, Levi S. Brown deserves special recognition for his observational skills and interpretive acumen. Brown worked for Lone Star Research Laboratory in Hudson, New York, in the 1930s and in 1940 joined the research staff at the Portland Cement Association, where he spent approximately 25 years in cement and concrete investigations. Most of his scientific efforts were dedicated to the microscopical interpretation of clinker burning, cement hydration, and concrete deterioration. An unpublished report (Brown, 1936) contains the following interesting observations:1. C3A and C3S were discriminated in thin sections and powders mounted in Hyrax, * a synthetic resin with index of refraction ** of approximately 1.710.2. Differences in optical properties of C3S were defined and birefringence and morphology were observed to show wide ranges; crystal zoning was not clearly understood.3. Optical characteristics of C2S, particularly the discrimination between polymorphic varieties (alpha, beta, and gamma), were described. The “better burned” clinkers were said to contain relatively clear crystals.4. Optical characteristics of C4AF, especially the color variations, were related to burning conditions, magnesium oxide content, and a reducing environment, the latter indicated by a honeybrown C4AF color and weak pleochroism. The darkening and strong pleochroism of C4AF were correctly thought to be due to incorporation of magnesium oxide.5. The morphologic and volumetric changes in the transformation of calcite to lime in a portland cement raw mix were described.6. Large crystals of periclase were described and explained as an effect of annealing of commercial clinkers.7. Gehlenite, found sparingly in practically all portland cement clinkers, was detected by examination of floating particles that have a uniaxial character and perfect basal cleavage in an oil of approximately 1.71 refractive index. Gehlenite was said to be suggestive of underburning.8. Sulfate minerals in clinkers, observed as floating particles in refractive index oil, were said to occur abundantly in underburned clinker. Optical characteristics of clinker sulfates compared with sulfate phases formed in the laboratory led to the conclusion that the low-index mineral in clinker is an alkali sulfate with a variable but small amount of calcium sulfate held in solid solution.9. Free lime was seen to increase with raw feed particle size and decrease with increasing burning time (flame length).
Brown and Swayze in 1938 published a paper describing the application of the microscope to autoclave problems, namely, free lime and magnesia in portland cement. Three forms of free lime were defined: (a) light-burned (quicklime), (b) hard-burned, and (c) air-slaked. The latter type was described as a “heretofore unidentified form of calcium hydroxide” having optical properties different from normal calcium hydroxide (portlandite) and thought to be the “Epezit” which was defined by Guttmann and Gille in 1928a and 1928b. Epezite was said to differ from portlandite in optical sign and refractive indices as follows:
| Epezite Uniaxial (+) | PortlanditeUniaxial (-) |
3. Microscopy of Cement Raw Mix and Clinker, Erling Fundal (1980, F.L. Smidth).As will be evident, the present writer has drawnheavily on the above three publications, plus several Japanese reports, particularly the work of Yoshio Ono (1995), whose detailed studies demonstrate the practicality of transmitted-light microscopy in the cement plant. Ono’s Method is discussed in Chapter 6.A commendable effort to bring cement and concrete microscopists together for the purposes of sharing knowledge and promoting the use of the microscope in the construction industry is seen in the founding of the International Cement Microscopy Association (ICMA) in 1980. * Published proceedings of their annual meetings have helped immeasurably in spreading knowledge of various microscopical methods and have generally stimulated growth in cement quality control through microscopy in North America.Illustrating quality-control methods in well cements, polarized-light microscopy and fluorescent microscopy have been applied to the analysis of oilwell cement blends containing pozzolans, bentonite, potassium chloride, friction reducer, modified pozzolan, fluid-loss addition, silica flour, and other materials (Reeves, Bailey, and Caveny, 1983). Examination of cement polished sections has shown a relationship of oil-well cement thickening times and retardation rates (Caveny, Weigand, and Bailey, 1983). Caveny and Weigand (1985) described a good oil-well cement as having well-formed alite (40 to 50 microns), no surficial deterioration of silicates, low free lime (less than 0.5%), and being free of metallic iron.Relatively recent contributions to oil-well cement microscopy include Polkowski (1987) who concluded that four cements with less than ideal microscopical characteristics still performed acceptably with different loadings of admixtures.Carruthers, Livesay, and Wells (1994) describe some of the burning conditions required for production of a Class H (HSR) oil-well cement: (1) hot burning zone and long retention time (dendritic belite), (2) high burning zone temperature and long burning zone (cannibalistic alite, wrap-around belite), (3) lengthening of burning zone and increasing temperature (belite beginning to disperse, silicate enlargement, and clarification of matrix), (4) dust recirculation (zoning in alite), slow cooling from extension of burning zone farther back in the kiln (ragged belite), and others. Desirable properties of the Class H (HSR) cement include a dead burned clinker with large alite, cannibalistic alite, amoeboidal belite, wrap-around belite, and finely crystalline C3A.
The desirable characteristics of Class H well cement were listed by Arbelaez (1990): free lime levels less than 0.5% with a uniform distribution, C 3A less than 6.5%, no weathered clinker, using only the 12.7- to 38.1-mm clinker fraction for the cement, relatively hot burning without production of cannibalistic alite, and avoidance of ragged belite by rapid cooling.The subject of clinker grindability also has microscopical aspects and the most complete literature survey, to date, is that of Hills (1995) who enumerated most of the prevailing agreed-upon relations (such as decreasing alite crystal size increasing grindability). Other variables on which the interpretations were not as clear cut (such as percent liquid phase) were also listed.Tachihata, Kotani, and Jyo (1981), in a laboratory study of the relationships between rate of heating, raw meal fineness, and other factors, concluded that clinkers with large crystal sizes in a narrow size range showed unfavorable grindability, and that cracks within the crystals and at the boundaries were some of the most important factors in grindability.Viggh (1994) studied clinker grindability and other related cement characteristics, concluding, among other things, that better grindability results with increase in liquid and alite percentages, and a decrease in alite crystal size. Poorer grindability resulted when belite percentage and crystal size increased. A decrease in setting time and improvement in strength development follow from better grindability. Cement flowage was said to be dependent on the amount of gypsum.Theisen (1993) described a rapid method of microscopical determination of alite and belite size and approximation of visible pore space by recording the number of intercepts along a line of traverse in successive fields of view. The intercept numbers were used with Bogue calculations and related to power consumption (kwh/t) in grinding. Data can be gathered in less than an hour.Many additional recent publications linking microscopy to a wide range of performance-related properties of cement are given in the following chapters