The Influence of Metakaolin on Limestone Reactivity in Cementitious Materials
Abstract
Recent studies have demonstrated that in the presence of limestone (CaCO3), carbonate-AFm phases (i.e., hemi- and/or mono-carboaluminate) may be stabilized at the expense of sulfate-AFm, which is more commonly found in cement systems. This suggests that enhancing AFm phase formation may be a novel way of incorporating increased quantities of limestone as a reactive component in cementbased systems. Often, in an ordinary portland cement (OPC), the quantity of the AFm hydrates formed is limited by the availability of aluminum. Therefore, as means of enhancing AFm phase formation, this paper evaluates metakaolin addition to determine how it affects limestone reactions and carbonate-AFm formation in the OPC systems. The results of a multi-method study including: X-ray diffraction with Rietveld refinement (QXRD), strength measurements, thermogravimetric analysis, and thermodynamic calculations are used to quantify solid phase constitutions, and the extent of limestone that has been consumed in reaction. Obtained results suggest that pozzolanic reactions which occur when metakaolin is used as an aluminous source are observed to be beneficial in offsetting the dilutive effects of OPC replacement noted in blended cement formulations.
Materials and Methods
An ASTM C150 compliant Type I/II ordinary portland cement (OPC) and commercially available limestone (nominally pure: >95 % CaCO3, d50 = 3 μm) were used. The oxide compositions of the OPC and metakaolin (MET) are presented in Table 1. A series of cementitious mixtures were prepared using de-ionized (DI) water at a fixed water-to-solids ratio (w/s = 0.45) as described in ASTM C305. The series of mixtures produced comprised of: (a) plain OPC, pastes in which: (b) 30 % of the OPC is replaced by limestone2 (c) 5-to-15 % of the OPC is replaced in 5 % increments by MET (d) 5-to-15 % of the OPC is replaced in 5 % increments by MET and an additional 30 % of the OPC is then replaced by limestone. Compressive strength measurements were carried out at 90 days using cubic specimens (50 mm × 50 mm × 50 mm) cured at 25 ± 1 °C in lime water as described in ASTM C109 [8]. For thermal analysis (TG/DTG) a Perkin Elmer STA 6000 thermal analyzer with a Pyris data acquisition interface was used to determine solid phase quantities in cementitious mixtures. To arrest hydration, solvent exchange was as described by Zhang and Scherer [13]. Quantitative X-ray diffraction analyses were carried out on powdered cementitious mixtures at desired ages using a Bruker D8 Advance diffractometer using Cu-Kα (λ = 1.54 Å) radiation. Care was taken to minimize preferred orientation errors by texturing the sample surface, and using a rotating sample stage. Xray structure information for the relevant anhydrous and hydrated crystalline phases was sourced from standard databases or from the literature [14, 15]. Rietveld analysis was carried out using the X’Pert HighScorePlus© [16]. Thermodynamic calculations were carried out using a geochemical speciation code, GEMS-PSI: version 2.3.1 [17]. Thermodynamic data of solid and aqueous species are sourced from the GEMS-PSI database, and amended with additional information relevant to cementitious systems [12]. Calculations were performed under conditions of 1 bar, 25 °C and CO2-free air.
Results and Discussion
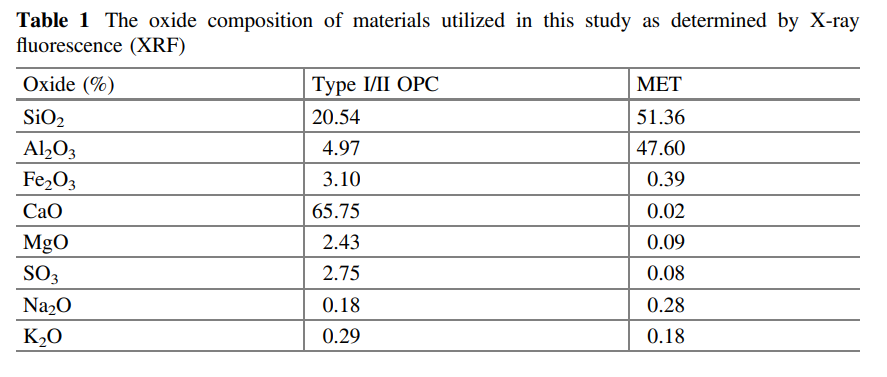
As showed on Fig. 1, the replacement of OPC by metakaolin results in improved
strength. Such beneficial effects of metakaolin addition, have also been highlighted
by Vance et al. [18, 19] and Antoni et al. [20] and attributed to the pozzolanic
character of metakaolin, and the formation of the Hc/Mc phases. This is significant
in that in spite of very substantial reductions in the cement factor (CF), mechanical
properties near equivalent to the pure OPC mixtures can be achieved. Figure 2
shows representative DTG traces at 90 days, and the portlandite content normalized
by the cement factor (CF) for metakaolin containing mixtures. It is noted that
portlandite contents of a given mixture decrease with increasing OPC replacement
by metakaolin, even when dilution is accounted for. This is indeed expected due to
the pozzolanic nature of metakaolin, which would consume lime to form a low(er)
Ca/Si (and potentially higher Al-substituted) C-S-H phase which ensures that metakaolin containing mixtures show reduced decreases in compressive strength, in
spite of the substantial OPC replacement. Figure 3 shows XRD patterns for the
metakaolin containing mixtures after 90 days of hydration. It is noted that the
portlandite content of a given mixture, in both limestone deficient/excess cases,
reduces with increasing metakaolin content. Mc exists as the dominant phase, only
when excess limestone is added to the system.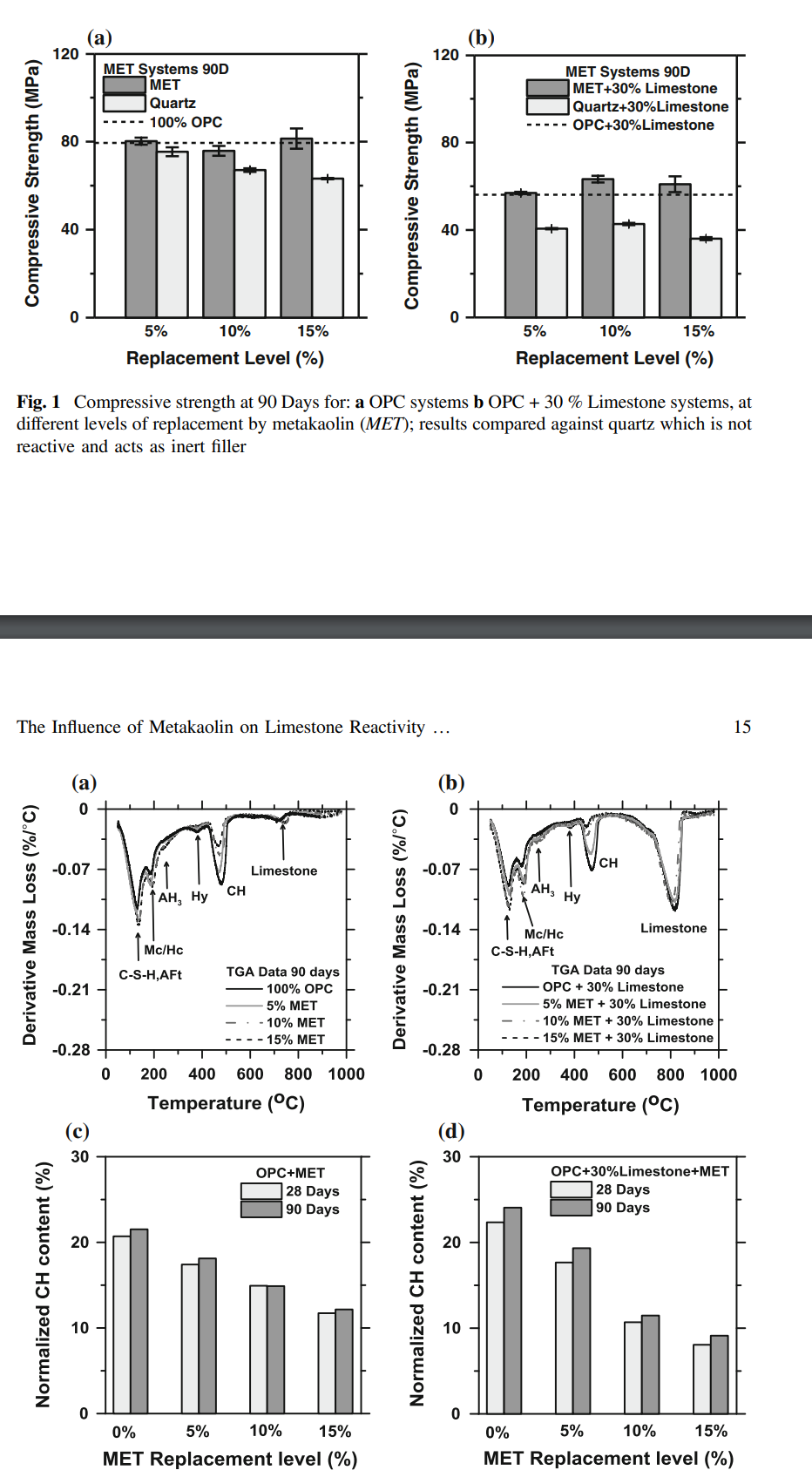
When only limestone intrinsic to the OPC (limestone deficient system) is
present, Mc is stabilized only when no metakaolin is added. However, when OPC is
replaced by metakaolin, in increasing proportion, Hc is stabilized as stable and
dominant phase. This is in line with the observations made by Antoni et al. [20] and
Vance et al. [18, 19]. Contents of the CO3-AFm (i.e., Hc/Mc) phases, reflect trends
in limestone consumption. More CO3-AFm phases form with increasing metakaolin
content, and when excess limestone is present. It should be noted that while these
trends follow evolutions in the equivalent CO2/Al2O3 ratio, limestone reaction is
limited, likely on account of its low dissolution rate. In spite of the near consumption of portlandite, e.g., in the 15 % metakaolin mixture containing excess
limestone, strӓtlingite is not observed in the XRD patterns. It was noted that ettringite is present in both the deficient and excess limestone cases, wherein the
release of sulfate ions, due to preferred CO3
2− ion uptake into the AFm phase,
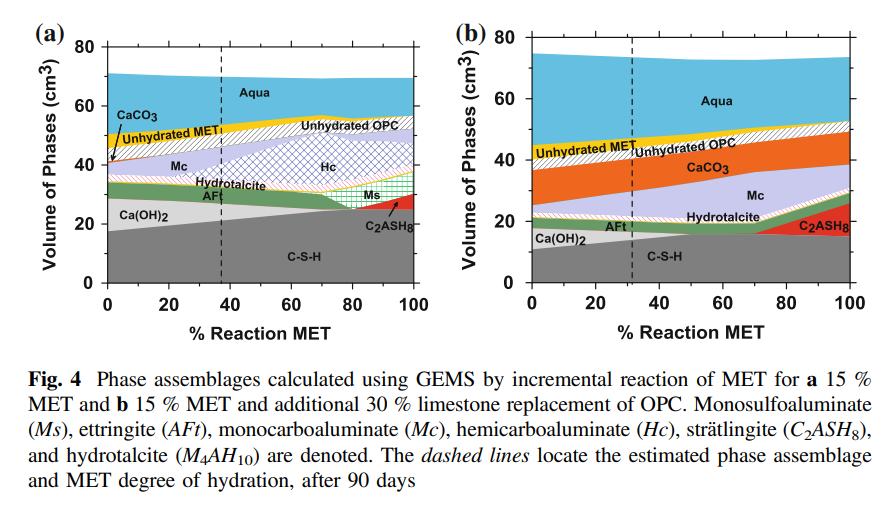
stabilizes ettringite in these systems. Figure 4 shows volumetric phase assemblages,
calculated using GEMS, for 15 % OPC replacement by metakaolin in either
limestone deficient or excess conditions. The systems presented show (determined
by the portlandite match-point from TGA data) a degree of metakaolin reaction of
37 and 31 %, in limestone deficient and excess scenarios respectively.
Due to pozzolanic reaction, metakaolin ensures consumption of lime and
increased formation of C-S-H formed, albeit, of a lower Ca/Si ratio and potentially
also higher Al substitution. In agreement with XRD data, both C2ASH8 and
Hydrogrossular phases are not predicted to form for relevant levels of metakaolin
reaction. In contrast to the observations herein, XRD data of Antoni et al. [20]
showed the presence of C2ASH8 and portlandite in coexistence with each other.
While this is in violation of thermodynamic phase relations established by Damidot
et al. [21], it may be on account of the somewhat higher metakaolin contents used in
their study, or inhomogeneous reaction zones. This discrepancy could however,
also be on account of uncertainties in determination of solubility data, small variations in which could alter stability fields dramatically. For example, as noted in the
simulations shown herein (Fig. 4), strӓtlingite is predicted to form only when
portlandite is consumed. When only limestone intrinsic to the OPC is present (see
Fig. 4a), at low levels of metakaolin reaction, Mc is stabilized. With the passage of
time and increase in metakaolin reaction the phase relation changes and Hc
becomes dominant with Mc existing as the minor CO3-AFm phase. For the degree
of metakaolin reaction ascertained herein, Mc and Hc appear to exist in equivalent
volumetric quantities, though on a mass basis this would translate to a larger
quantity of Mc (the denser CO3-AFm phase), in contrast to the XRD data shown in
Fig. 3. While this may indicate that the ascertained degree of metakaolin reaction is
lower than reality, it may also suggest kinetic restraint in the formation of Hc, from
the Mc, Ms and portlandite (or vice versa depending on the chemical environment).
When excess limestone is present, Mc is predicted to be the dominant AFm
phase (Fig. 4b). This is once again in contrast to the XRD observations in Fig. 3,
where even under limestone excess conditions, when (15 %) metakaolin is present,
Hc and Mc are noted to coexist; the reasoning for which is discussed above. It is
seen that, at any degree of metakaolin reaction, a large quantity of limestone
remains unreacted, ensuring modest levels of Hc/Mc formation. This leads to the
idea that the compensation in mechanical properties (see Fig. 1), produced by the
combined replacement of OPC, by limestone and metakaolin, is dominantly on
account of the pozzolanic nature of metakaolin and less due to the formation of
CO3-AFm phases.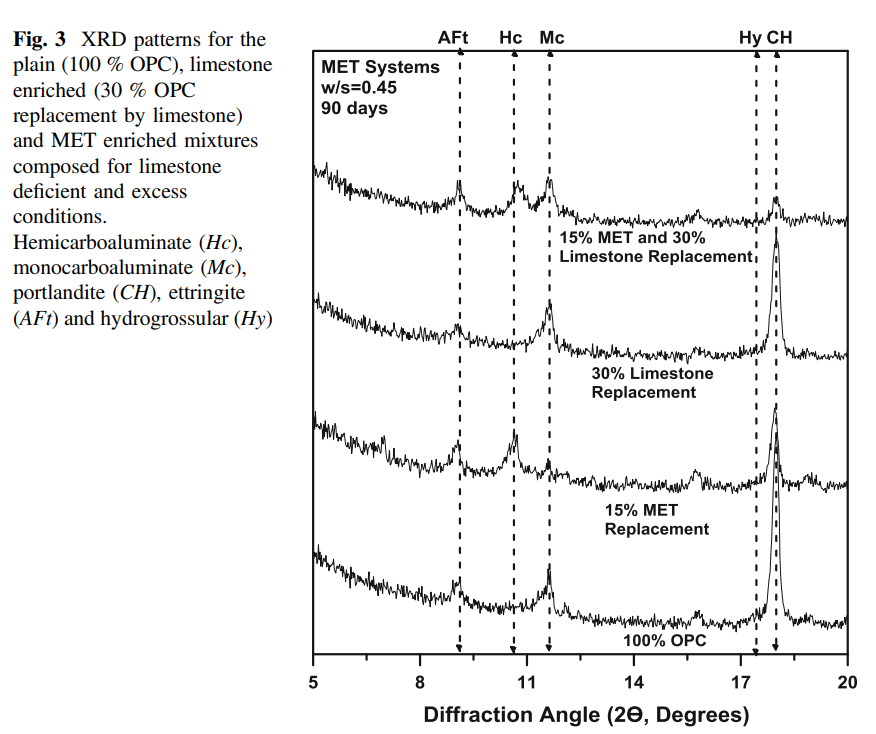
Conclusions
Metakaolin was evaluated in blended binder formulations in terms of its ability to enhance the reaction of limestone in cementitious systems. Such enhancements in limestone reaction are provoked by systematic manipulation of the binder chemistry, e.g., in terms of the SO3/Al2O3 and CO2/Al2O3 ratios of the binder. It is noted that in spite of the provision of sufficient aluminium, calcium and water, the extent of limestone reacted is limited, mostly on account of its low reactivity (i.e., dissolution rate, and solubility). Due to reasonable (reactive) aluminum content metakaolin has shown to increase limestone reactivity and resulting formation of the CO3-AFm (hemi- and/or mono-carboaluminate). On the account of its pozzolanic nature, it is also effective at ensuring strength equivalence, or improvement (in spite of reductions in the OPC content) as compared to the pure OPC formulations.